A new meta-analysis has confirmed the suspicions of many clinicians about the relative effects of sugammadex and neostigmine on adverse effects and turnover rates following the reversal of neuromuscular blockade.
The study found that use of sugammadex facilitated faster extubation time, OR turnover time and PACU discharge time, and was also associated with lower risk for bradycardia and pneumonia compared with neostigmine.
“Sugammadex is a newer, widely used reversal agent, but some studies still cite harmful effects with its use,” said Javier Marull, MD, an associate professor of anesthesiology and pain management at The University of Texas Southwestern Medical Center, in Dallas. “Given its rapid rise in usage, a more comprehensive characterization of the clinical and practical aspects of sugammadex compared to the standard of neostigmine is needed.”
To do so, Marull and his colleagues systematically searched the PubMed and Scopus databases for publications comparing the efficacy and safety of sugammadex with those of neostigmine. All publications that included either end point were included, regardless of publication date, country of origin, language, age range of patients, type of surgical procedure or patients’ American Society of Anesthesiologists physical status.
As reported in a presentation to the 2022 virtual annual meeting of the International Anesthesia Research Society, Association of University Anesthesiologists and Society of Critical Care Anesthesiologists (abstract 1656), Marull noted that the search yielded 248 randomized controlled trials and articles. Of these, 57 articles were included in the final analysis, comprising 66,157 patients. It was found that compared with neostigmine, sugammadex demonstrated significant reductions in several end points, including:
- extubation time (mean difference, –2.77 minutes; 95% CI, –3.95 to –1.59 minutes),
- recovery to train of four (TOF) greater than 0.9 (mean difference, –11.27 minutes; 95% CI, –12.7 to –9.89 minutes),
- OR discharge time (mean difference, –3.74 minutes; 95% CI, –4.77 to –2.71 minutes), and
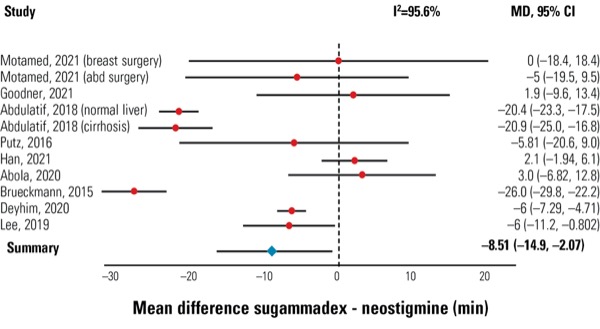
- PACU discharge time (mean difference, –8.51 minutes; 95% CI, –14.9 to –2.07 minutes; Figure 1).
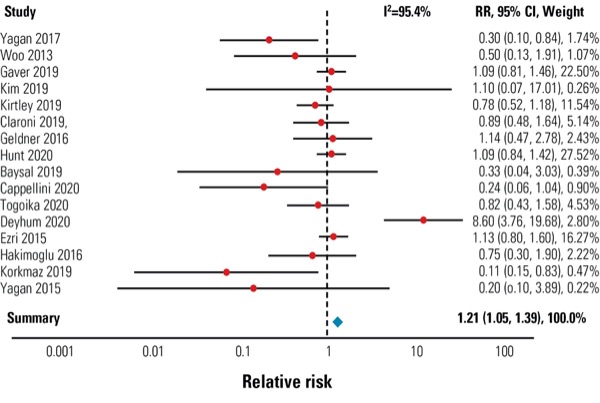
Figure 2. Relative risk for postoperative nausea and vomiting when sugammadex versus neostigmine is administered.
For adverse events, the meta-analysis revealed that use of sugammadex resulted in significant reductions in both pneumonia (relative risk [RR], 0.593; 95% CI, 0.361-0.671) and bradycardia (RR, 0.535; 95% CI, 0.424-0.675). In contrast, significant increases in postoperative nausea and vomiting were associated with sugammadex use relative to neostigmine (RR, 1.21; 95% CI, 1.05-1.39) (Figure 2). Finally, no significant differences were found between neostigmine and sugammadex in occurrence of atelectasis (RR, 0.964; 95% CI, 0.853-1.09).
“We were surprised at the increased rate of postoperative nausea and vomiting with sugammadex, but we can tailor our anesthetic plan to focus more on managing and preventing sugammadex-related postoperative nausea and vomiting,” Marull said in an interview with Anesthesiology News.
As the researchers concluded, the study supports the use of sugammadex as a reversal agent for neuromuscular blockade.
“Because of its safer adverse-effect profile, easier administration and quicker turnaround time, sugammadex is preferred over neostigmine in patients who are not at risk for intractable nausea and vomiting,” Marull said.
The researchers are currently performing a cost–benefit analysis of sugammadex’s superior turnover rates and adverse-effect profile relative to neostigmine. “These results serve as a strong basis for future work on neuromuscular blockage reversal agents, with large implications in improving the quality of patient care, bolstering the efficiency of the surgery and anesthesiology services, as well as improving healthcare costs of surgery and anesthesia,” the authors noted.
For Stephan Thilen, MD, MS, one of the primary drawbacks of the meta-analysis is that it is based on published studies that have largely focused on reversal from a depth of neuromuscular blockade that is too deep for neostigmine to be effective. “To date, there has been only one randomized controlled trial comparing sugammadex to neostigmine for reversal of a level of block that we now refer to as minimal [TOF ratio, 0.4 to =0.9],” commented Thilen, an associate professor of anesthesiology and pain medicine at the University of Washington School of Medicine, in Seattle. “The authors of that study found that sugammadex and neostigmine both reversed a block with TOF ratio of 0.5 in a comparable manner.
“In a mixed surgical population and without excessive use of muscle relaxants, minimal block is quite common at the end of surgery,” Thilen continued. “We have found in our own studies that [30% to] 40% of patients have this level of block. They can be reversed effectively with a small dose of neostigmine, which often costs approximately 90% less than sugammadex.”
As Thilen also pointed out, routine quantitative monitoring is vital to effectively reversing neuromuscular blockade. “Only with quantitative monitoring are we able to identify the patients who have spontaneously recovered to a TOF ratio equal to or greater than 0.9; these patients should not be exposed to the risk and cost of pharmacological reversal drugs,” he noted. “I agree that for levels of block that are deeper than minimal, sugammadex is the preferred drug. But failing to account for the depth of block in a meta-analysis is a notable shortcoming.”
—Michael Vlessides
Marull and Thilen reported no relevant financial disclosures.